Thus, the area enclosed by the cycle on the T-s diagram also represents the net work done by the cycle. By the first law applied to the cycle, the net heat added to the cycle is equal to the net work done by the cycle. Can this result be used to improve the cycle efficiency? What happens to th, win /wout, and wnet as the pressure ratio rp is increased? Consider the T-s diagram for the cycle and note that the area enclosed by the cycle is the net heat added to the cycle. The back work ratio is defined as Note that T4 = 659.1 K > T2 = 492.5 K, or the turbine outlet temperature is greater than the compressor exit temperature. The net work done by the cycle is The cycle efficiency becomes Since P3 = P2 and P4 = P1, we see that We have already shown the heat supplied to the cycle per unit mass flow in process 2-3 is The conservation of energy for the turbine, process 3-4, yields for constant specific heats (let’s take a minute for you to get the following result) Since process 3-4 is isentropic The conservation of mass givesįor constant specific heats, the compressor work per unit mass flow is Since the compressor is isentropic Apply the conservation of energy for steady-flow and neglect changes in kinetic and potential energies to process 1-2 for the compressor. Determine the compressor work and the turbine work per unit mass flow, the cycle efficiency, the back work ratio, and compare the compressor exit temperature to the turbine exit temperature. The pressure ratio rp is 6:1 and the air leaves the heat addition process at 1100 K. 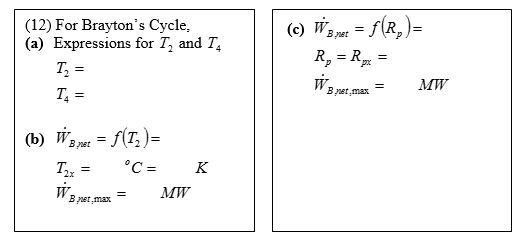
Note that this approach does not require the closed cycle assumption.Įxample 9-2 The ideal air-standard Brayton cycle operates with air entering the compressor at 95 kPa, 22oC. Compare your result with the above expression. Where the pressure ratio is rp = P2/P1 and Extra Assignment Evaluate the Brayton cycle efficiency by determining the net work directly from the turbine work and the compressor work. Recall processes 1-2 and 3-4 are isentropic, so Since P3 = P2 and P4 = P1, we see that The Brayton cycle efficiency becomes Is this the same as the Carnot cycle efficiency? Since process 1-2 is isentropic, The conservation of energy for process 4-1 yields for constant specific heats (let’s take a minute for you to get the following result) The thermal efficiency becomes The conservation of mass gives For constant specific heats, the heat added per unit mass flow is Apply the conservation of energy to process 2-3 for P = constant (no work), steady-flow, and neglect changes in kinetic and potential energies. Thermal efficiency of the Brayton cycle Now to find Qin and Qout. Process Description 1-2 Isentropic compression (in a compressor) 2-3 Constant pressure heat addition 3-4 Isentropic expansion (in a turbine) 4-1 Constant pressure heat rejection The T-s and P-v diagrams are We assume the working fluid is air and the specific heats are constant and will consider the cold-air-standard cycle. Therefore, an open system, steady-flow analysis is used to determine the heat transfer and work for the cycle. This cycle differs from the Otto and Diesel cycles in that the processes making the cycle occur in open systems or control volumes. Brayton Cycle The Brayton cycle is the air-standard ideal cycle approximation for the gas-turbine engine.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
